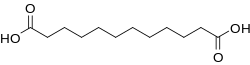
Dodecanedioic acid
| |
| Names | |
|---|---|
| Preferred IUPAC name
Dodecanedioic acid | |
| Other names
DDDA
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.010.680 |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C12H22O4 | |
| Molar mass | 230.304 g·mol−1 |
| Appearance | White flakes |
| Density | 1.066 g/cm3 |
| Melting point | 127–129 °C (261–264 °F; 400–402 K) |
| Boiling point | 245 °C (473 °F; 518 K) |
| pH dependent | |
| Hazards | |
| Flash point | 220 °C (428 °F; 493 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Dodecanedioic acid (DDDA) is a dicarboxylic acid with the formula (CH2)10(CO2H)2. A white solid, the compound finds a variety of applications ranging from polymers to materials. The unbranched compound is the most commonly encountered C12 dicarboxylic acid.
Production
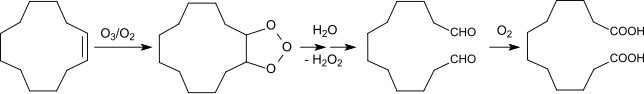
[edit]DDDA has traditionally been produced from butadiene using a multi-step chemical process.[1] Butadiene is first converted to cyclododecatriene through cyclotrimerization.[2] The triene is then hydrogenated to cyclododecane. Autoxidation by air in the presence of boric acid gives a mixture of cyclodecanol and the cyclododecanone. In the final step, this mixture oxidized to the diacid using nitric acid. An alternative route involves ozonolysis of cyclododecene.[3]
Biological process
[edit]Paraffin wax can be converted into DDDA on a laboratory scale[4] with a special strain of Candida tropicalis yeast in a multi-step process.[5] Renewable plant-oil feedstocks sourced from switchgrass could also be used to produce DDDA.[1]
Uses
[edit]DDDA is used in antiseptics, top-grade coatings, painting materials, corrosion inhibitors, surfactants, and polymers. It is one of two precursors to the engineering plastic nylon 612.[6] The once commercial nylon called Qiana was produced on scale using DDDA. DDDA ester with ethylene glycol is a synthetic musk of the macrocyclic lactones group commercially marketed as "Arova 16".
Medical
[edit]In type 2 diabetic patients DDDA demonstrated that IV infusion helps to maintain normal blood sugar and energy levels without increasing the blood glucose load in the process.[7]
References
[edit]- ^ a b "BIOLON® DDDA". verdezyne.com. Archived from the original on 2016-09-24. Retrieved 2016-09-23.
- ^ Klaus Weissermel, Hans-Jurgen Arpe (1997). Industrial Organic Chemistry (3rd ed.). John Wiley & Sons. ISBN 3-527-28838-4.
- ^ Cornils, Boy; Lappe, Peter; By Staff, Updated (2014). "Dicarboxylic Acids, Aliphatic". Ullmann's Encyclopedia of Industrial Chemistry. pp. 1–18. doi:10.1002/14356007.a08_523.pub3. ISBN 9783527306732.
- ^ "Dibasic acids". www.cathaybiotech.com. Archived from the original on 2018-10-09. Retrieved 2019-03-15.
- ^ Kroha, Kyle. "Industrial biotechnology provides opportunities for commercial production of new long-chain dibasic acids" (PDF). Inform. 15(9) (Sep 2004). American Oil Chemists Society: 568. Archived from the original (PDF) on 6 October 2014. Retrieved 15 March 2019.
- ^ Nylon#Homopolymers
- ^ Greco, A. V.; Mingrone, G; Capristo, E; Benedetti, G; De Gaetano, A; Gasbarrini, G (1998). "The metabolic effect of dodecanedioic acid infusion in non-insulin-dependent diabetic patients". Nutrition. 14 (4): 351–7. doi:10.1016/s0899-9007(97)00502-9. PMID 9591306.